How Do You Know How Many Valence Elctrons an Element Has
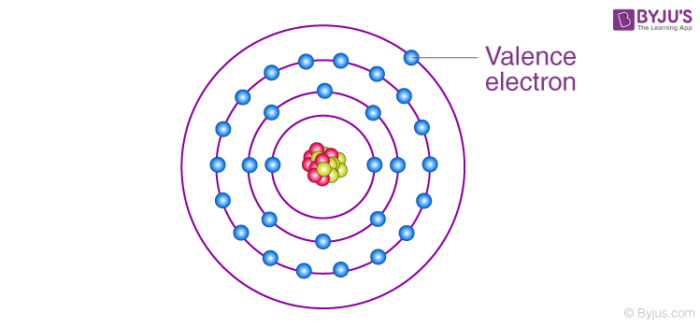
Valence electrons are the s and p electrons in the outermost beat out. The electrons present in the inner shell are cadre electrons. When nosotros study and discover the atom of an element, nosotros come across tiny subatomic particles called valence electrons. Lewis structures help the states to runway the valence electrons and predict the types of bond.
Valence electrons are all arranged in different orbitals or shells and are mostly negatively charged particles. Farther, these electrons are responsible for interaction between atoms and the formation of chemic bonds. Yet, not all electrons are associated with the cantlet. Only the electrons present in the outermost beat can participate in the formation of a chemical bond or a molecule. Such type of electrons is chosen valence electrons.
Tabular array of Contents
- What are Valence Electrons?
- Related Videos
- Characteristics of Valence Electron
- Determination of Valence Electrons
- Valence Electron of Elements
- Frequently Asked Questions – FAQs
What are Valence Electrons?
Valence is the number of electrons an atom must lose or proceeds to attain the nearest noble gas or inert gas electronic configuration. "Electrons in the outer shells that are not filled are called valence electrons" .
The valence electrons are function of most of the chemical reactions considering they contain more than energy compared to the electrons present in inner orbits. Meanwhile, the number of valence electrons present also helps usa determine a specific element's chemical backdrop, such every bit its valence or valency, the formation of bonds with other elements. It besides gives us an idea of how readily the atoms tin form bonds, the number of unpaired electrons and how many atoms can take office.
Related Videos

Characteristics of Valence Electron
Electrons are involved in the chemical bonding and reactions of the atom. It is said to occupy orbitals in an atom. The number of valence electrons of an atom can be obtained from the periodic table considering it is equal to the group number of the atom. Atoms are most stable if they have a filled valence crush of electrons. Atoms transfer or share electrons in such a way that they can accomplish a filled crush of electrons.
Some key characteristics of a valence electron are;
- For the primary group elements, the valence electron exists only in the outermost electron beat out.
- A valence electron tin can be in the inner shell of a transition metallic.
- An cantlet consisting of a airtight shell of valence electrons volition usually be chemically inert.
- A valence electron can either blot or release energy in the form of a photon.
- Valence electrons as well determine the conductivity of an chemical element. Depending on in this nature of elements tin can be a metal, non-metal, or a metalloid.
Conclusion of Valence Electrons
One of the easiest ways to find valence electrons is past checking out the elements' place in the periodic table. The valence electrons of an element can be found out past closely examining the vertical column in which the elements are grouped in. By looking at the group number that is given we can identify the number of valence electrons that an element which is listed under that specific column has.

Another way to notice or decide valence electrons is by knowing the electronic configuration.
However, if we take the transition metals (groups 3-12), finding the valence electron is quite complicated. These elements atomic construction is rigid and the d subshell is incomplete and sits lower than the outer shell.
Valence Electron of Elements
Here is a list of the number of valence electrons present in the different groups.
| Periodic Tabular array Grouping | Valence Electrons |
| Alkali metals – Group 1 (I) | one |
| Alkaline world metals – Group ii (2) | 2 |
| Boron group – Group 13 (3) | iii |
| Carbon group – Group 14 (Iv) | 4 |
| Nitrogen group – Grouping 15 (V) | 5 |
| Oxygen group – Group 16 (Half dozen) | 6 |
| Halogens – Group 17 (VII) | 7 |
| Noble gases – Group eighteen (Viii or 0) | viii |
Often Asked Questions – FAQs
How do you find the valence electrons?
For neutral atoms, the number of valence electrons is equal to the atom's primary group number. The main group number for an chemical element tin can be found from its cavalcade on the periodic tabular array. For case, carbon is in group 4 and has iv valence electrons. Oxygen is in group 6 and has six valence electrons.
What is valence electron with case?
The total number of electrons present in last crush orbit is known equally valence electron. For example: Oxygen have vi electrons present in last orbital shell hence 6 is valence electron.
What is called valency?
Valency is the combining ability of an chemical element. Elements in the same group of the periodic table accept the same valency. The valency of an element is related to how many electrons are in the outer shell.
What is the purpose of valence electrons?
Valence electrons are the outer-shell electrons of an atom. Valence electrons determine the reactivity of an cantlet.
How practise valence electrons work?
Valence electrons are the electrons located at the outermost shell of an atom. … Because when two atoms interact, the electrons in the outermost shells are the first ones to come into contact with each other and are the ones that determine how an atom will react in a chemic reaction.
To learn more about this topic and other related topics, annals with BYJU'Due south and download the mobile awarding on your smartphone.
Source: https://byjus.com/chemistry/valence-electrons/
0 Response to "How Do You Know How Many Valence Elctrons an Element Has"
Post a Comment